When you’re on a combination of NTI drugs, even a tiny change in dosage can be dangerous. These aren’t ordinary medications. They sit on a razor’s edge - too little and they don’t work; too much and they can kill you. Now imagine your pharmacy swaps the brand-name version for a generic. Sounds harmless, right? For most drugs, yes. For combination NTI therapy? It’s a gamble with your life.
What Exactly Are NTI Drugs?
Narrow Therapeutic Index (NTI) drugs have almost no room for error. The difference between a therapeutic dose and a toxic one is razor-thin. Think warfarin, levothyroxine, lithium, digoxin, or phenytoin. One pill too much, and you bleed internally. One pill too little, and your clot forms unchecked. These drugs require constant monitoring - blood tests, dose tweaks, careful tracking. That’s why they’re not taken casually.
The FDA defines NTI drugs by five key traits: minimal separation between effective and toxic levels, high risk of life-threatening failure or side effects, need for regular blood monitoring, low variability within the same person, and frequent small dose adjustments. These aren’t theoretical concerns. They’re daily realities for patients managing atrial fibrillation, thyroid disorders, epilepsy, or heart failure.
Why Combination NTI Therapy Is So Risky
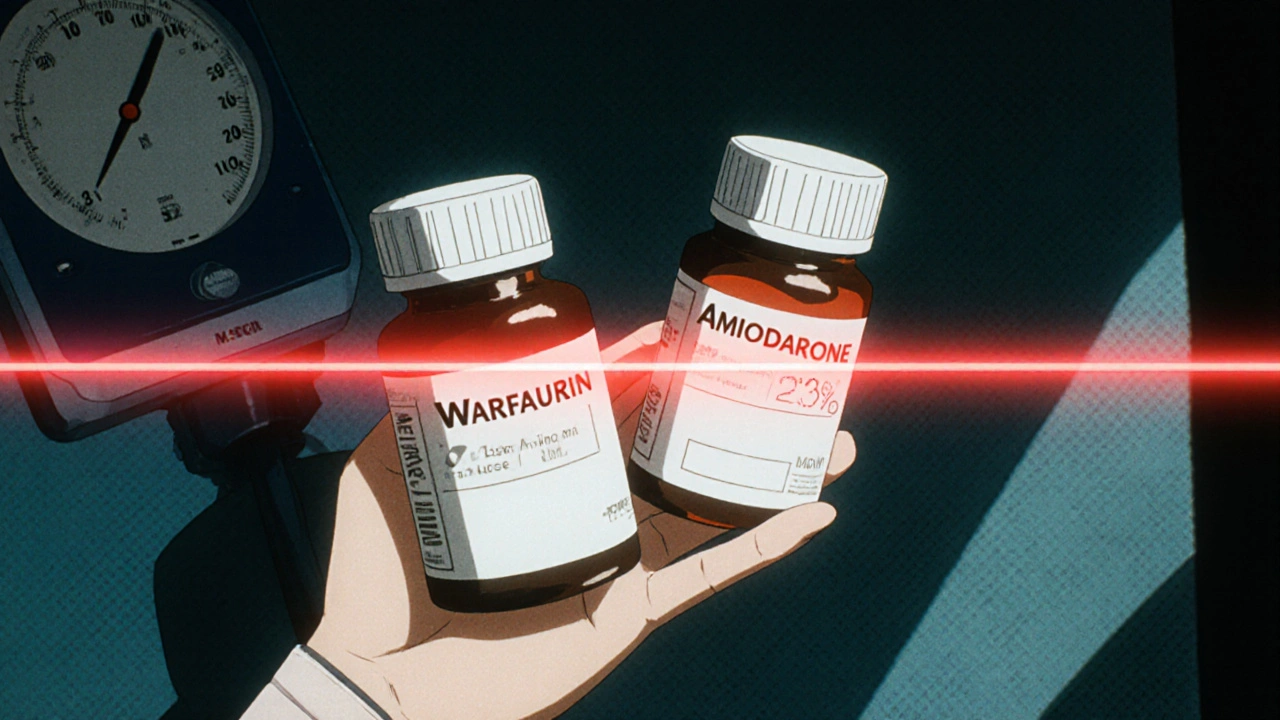
Combination therapy means using two or more NTI drugs together. This happens in complex cases - like a patient on warfarin and amiodarone for heart rhythm control, or someone on methotrexate and another targeted agent for leukemia. The idea is to hit the disease from multiple angles. But here’s the catch: when you combine two drugs that each have a 10% margin for error, you don’t get a 20% margin. You get a compounded risk.
Let’s say Drug A has a 90-111% bioequivalence window. Drug B does too. When combined, the total variation can swing up to 22% - meaning your blood levels could be 22% higher or lower than expected. That’s not a minor fluctuation. That’s enough to send your INR from 2.5 to 6.8 in three days, triggering a hospital visit. A 2020 JAMA Internal Medicine study found patients on combination therapies with even one NTI drug had a 27% higher rate of adverse events after generic substitution compared to just 8% in non-NTI combinations.
Why There Are Almost No Generic Combination NTI Drugs
You can buy generic warfarin. You can buy generic levothyroxine. But you can’t buy a pill that combines warfarin and amiodarone - because no generic version exists in the U.S. market. Not one.
The reason? Bioequivalence standards. For regular drugs, generics must match the brand within 80-125% for absorption. For NTI drugs, that tightens to 90-111% for peak levels and 90-112% for total exposure. But when two NTI drugs are in one pill? The FDA says that’s not enough. In 2023, they proposed even stricter standards: 90-107.69% for Cmax and 90-110% for AUC. That’s a massive leap in manufacturing precision. Most companies can’t hit it. The cost of testing is astronomical. The risk of failure is too high.
Compare that to non-NTI combinations like entecavir/tenofovir for HIV - dozens of generics exist. But for NTI combos? The FDA Orange Book shows only 12.6% of combination regimens involving NTI drugs have generic alternatives. The rest? Still locked behind brand-name prices.

Real-World Consequences
Patients aren’t just statistics. They’re people.
A Reddit user on r/medicine posted in June 2023: “After my pharmacy switched to generic warfarin, my INR went from 2.5 to 6.8 within 3 days. I ended up in the ER. Now imagine if both drugs were generic.” That’s not an outlier. A 2022 Drugs.com survey of 1,247 patients on NTI combinations found 63.4% had adverse effects after generic substitution - compared to just 18.2% on brand-name versions.
Pharmacists are seeing it too. A 2023 ASHP survey of 856 pharmacists revealed 78.3% had witnessed therapeutic failure after generic substitution in NTI-containing combinations. Over 40% reported serious adverse events - bleeding, seizures, heart arrhythmias. One pharmacist in Ohio told me: “I’ve had to call doctors at 2 a.m. because a patient’s lithium level spiked after a switch. It’s not a theoretical risk. It’s daily work.”
Even monitoring doesn’t fully fix it. Stabilizing a patient on a combination NTI regimen takes 6-8 weeks. They need 3.2 dose changes on average. For non-NTI drugs? Just 1.8. The cost? $1,200 to $2,500 a year in lab tests - double what non-NTI patients pay.
Who’s Speaking Up?
The American Society of Health-System Pharmacists (ASHP) doesn’t mince words. Their 2021 position statement says automatic substitution of generic NTI drugs in combination regimens poses an “unacceptable risk of therapeutic failure.” They’ve urged pharmacies and insurers to block automatic switches.
Dr. Janet Woodcock, former head of the FDA’s Center for Drug Evaluation and Research, said in 2022: “NTI drugs require special consideration. Combination products? They present even greater scientific challenges.”
But not everyone agrees. Generic manufacturers like Teva and Sandoz point to Europe. Since 2015, generic levothyroxine combinations have been used there with less than 2% adverse event rates. They argue modern manufacturing can deliver the precision needed.
Then there’s Dr. Donald Berry, who wrote in Nature Reviews Drug Discovery: “The 90-111% window still allows 22% total variation. When you combine two such drugs, that’s not just noise - it’s clinical danger.”
The Gap Between Access and Safety
The global NTI drug market is worth $48.7 billion. But combination NTI products? Less than 0.3% of that. Why? Because the science hasn’t caught up to the need.
Patients with complex conditions - cancer, severe arrhythmias, autoimmune disorders - need these combinations. But they’re priced out. Brand-name combos can cost $5,000 a month. Generic monotherapies? $10 to $50. The gap isn’t just financial. It’s therapeutic. Patients are stuck between unaffordable brand-name combos and dangerous generic substitutions.
Only 12 of 50 major U.S. academic medical centers have specialized NTI combination clinics. Most patients are managed by general pharmacists who haven’t had the 120+ hours of training ASHP recommends. Documentation for these combos? Poor. FDA-approved NTI monotherapies score 4.2 out of 5 on clarity. Combination products? Just 2.8.
What’s Next?
The FDA is working on a 2024 pilot program for “precision bioequivalence” testing using pharmacometric modeling - essentially, computer simulations of how drugs behave in the body. It’s promising. But it’s years away from routine use.
Meanwhile, insurers and pharmacy benefit managers keep pushing for cost savings. Automatic substitution rules still apply in many states unless explicitly blocked. And without federal policy changes, pharmacists are caught in the middle: pressured to save money, but legally responsible when something goes wrong.
Some experts, like Dr. Robert Temple of the FDA (ret.), believe manufacturing advances will eventually close the gap. Others, like Dr. Lewis Nelson of NYU, argue the problem is fundamental: “Combining multiple narrow-window drugs creates a pharmacokinetic nightmare. We may never achieve true therapeutic equivalence with current technology.”
The truth? We’re stuck. We have patients who need these drugs. We have generics that could make them affordable. But we don’t yet have the science, the standards, or the safety net to make it work.
What Patients and Providers Can Do Today
- Ask your doctor: Is this a combination NTI regimen? If yes, insist on brand-name unless you’re under close monitoring.
- Never allow automatic substitution without explicit consent from your prescriber.
- Get your blood tested regularly - and keep records. Track your INR, lithium levels, or other key markers.
- Speak up to your pharmacist. If they try to switch your meds, ask: “Is this safe for my combination?”
- Join patient advocacy groups. Your voice matters in pushing for better standards.
There’s no easy fix. But ignoring the problem isn’t an option. For patients on combination NTI therapy, the stakes aren’t theoretical. They’re life or death.
What does NTI mean in drug terms?
NTI stands for Narrow Therapeutic Index. It means the difference between a safe, effective dose and a toxic one is very small. Even minor changes in blood levels can cause serious side effects or treatment failure. Examples include warfarin, lithium, and levothyroxine.
Why are generic combination NTI drugs so rare?
Because proving bioequivalence is nearly impossible. When two NTI drugs are combined, even tiny differences in absorption between the brand and generic versions can compound. The FDA requires much tighter standards (90-111% for Cmax and AUC) than for regular drugs, and even those may not be enough. Most manufacturers can’t meet the cost or precision needed.
Can I switch from brand to generic for my NTI combination?
It’s risky. Studies show 27% higher adverse event rates when generics are substituted in combination NTI therapy. Only switch under direct supervision of your doctor with frequent blood tests. Never allow automatic substitution - insist on a manual override.
Are there any safe generic NTI combinations?
Very few. In the U.S., there are no FDA-approved fixed-dose combinations of two NTI drugs. In Europe, some levothyroxine-based combos have been used safely with strict monitoring, but these are exceptions. Most combination NTI therapies still rely on brand-name products.
What should I do if my pharmacy switches my NTI meds?
Contact your doctor immediately. Get a blood test within 3-5 days. Check your INR, lithium level, or other relevant markers. If levels are outside your target range, ask for the original brand. Document everything. You have the right to refuse substitution for NTI drugs.
Is there any progress being made on this issue?
Yes. The FDA proposed stricter bioequivalence standards in 2023 and plans a pilot program in 2024 using computer modeling to better predict how combination NTI drugs behave. But these are still in development. Don’t expect widespread availability of safe generics anytime soon.

james lucas
November 25, 2025 AT 11:25man i had no idea this was such a wild ride until my buddy got switched to generic warfarin and ended up in the er with an inr of 7.2. like, i thought generics were just cheaper versions of the same thing, but turns out when you got two drugs that are already on a knife’s edge, mixing em in a pill? total gamble. i mean, even the pharmacists are sweating bullets about it. no wonder people are getting scared to switch.
Michael Fitzpatrick
November 26, 2025 AT 11:32it’s wild how the system pushes for savings but ignores the human cost. i work in a clinic and we’ve had three patients in the last year with serious bleeds after a generic switch. none of them had a choice. the pharmacy did it automatically. the worst part? they all had to pay out of pocket for the brand again. it’s not just about money - it’s about trust. if your meds can randomly turn deadly, how do you even sleep at night?
Danny Nicholls
November 26, 2025 AT 15:22bro this is insane 😭 i’ve been on levothyroxine + lithium for years and my doc literally told me never to let them switch me. i used to think it was just paranoia but now i read this and it’s like… holy crap. i had no idea the variation could be 22% when combined. i’ve been doing monthly labs just to be safe. if you’re on combo ntis, don’t let anyone touch your script without your signature. and if your pharmacy tries to auto-switch? tell em to call your doc. i’ve done it twice and they backed off. you got rights, people 🙌
Robin Johnson
November 27, 2025 AT 12:22the fact that we’re still letting pharmacists make these decisions without explicit consent is a failure of the entire system. patients aren’t lab rats. if you’re on two ntis, your doctor should have to sign off on every substitution. period. the data is clear - adverse events jump 27%. we’re not talking about a headache here. we’re talking about strokes, seizures, death. if your insurer won’t cover the brand, that’s their problem. not yours. stand your ground.
Rahul Kanakarajan
November 28, 2025 AT 22:10why are people making such a big deal? in india we use generics for everything and no one dies. you just monitor and adjust. this is just american overreaction. if your body can’t handle a small variation, maybe you’re not the patient you think you are.
New Yorkers
November 29, 2025 AT 17:43we’re not just talking about medicine here. we’re talking about the death of compassion in healthcare. the system doesn’t care if you live or die - it cares if you’re profitable. these drugs? they’re not pills. they’re lifelines. and someone’s trying to cut them in half to save 50 bucks. i’m not mad. i’m just… heartbroken. this isn’t capitalism. this is negligence dressed up as efficiency.
David Cunningham
November 30, 2025 AT 13:24australia’s been doing combo ntis with generics for a while now and honestly? we’ve got a solid registry system. every switch gets logged, every blood test tracked. it’s not perfect, but it works. maybe the us needs better infrastructure, not just stricter rules. we don’t need to ban generics - we need better monitoring. and yeah, maybe a little less bureaucracy.
luke young
December 2, 2025 AT 03:10really appreciate this post. i’ve been on warfarin + amiodarone for 5 years and i never knew the combo was this fragile. i’ve always asked for the brand, but now i’m gonna start documenting every interaction with my pharmacy. maybe i’ll even write my rep. if enough of us speak up, maybe they’ll listen. thanks for the clarity.
Jessica Correa
December 2, 2025 AT 19:51my mom switched to generic levothyroxine and her heart started racing. she didn’t tell anyone for a week because she didn’t want to be a bother. by the time she went in, her tsh was off the charts. now she’s back on brand and she’s fine. but what if she hadn’t noticed? what if she’d been on two of these? this isn’t theoretical. it’s happening to real people every day.
manish chaturvedi
December 3, 2025 AT 07:37as someone from india who now works in a u.s. hospital, i see the contrast clearly. here, the fear of liability prevents progress. there, the lack of resources forces adaptation. neither is ideal. but perhaps the solution lies not in banning generics, but in building robust pharmacovigilance systems - real-time monitoring, patient registries, mandatory reporting. innovation should serve patients, not just profits.